Awiqli (Insulin Icodec): The Once-Weekly Insulin Changing Diabetes Forever
Introduction
More than 537 million adults worldwide live with diabetes — and that number is rising every year (WHO, 2021). For millions of people with type 2 diabetes who need basal insulin, daily injections have long been the only option. That changes now. Awiqli (insulin icodec-abae), developed by Novo Nordisk, is the world’s first FDA-approved once-weekly basal insulin, approved on 27 March 2026. This long-acting insulin reduces injections from seven to just one per week.
Furthermore, Awiqli belongs to the class of long-acting basal insulin analogues. However, it works very differently from daily insulins like glargine or degludec. In addition, it is already approved in the EU, Canada, Australia, Japan, and 13 other countries. In this guide, we explore how Awiqli works, its clinical benefits, correct dosage, and side effects.
What Is Type 2 Diabetes?
Type 2 diabetes (T2D) is a chronic condition where the body cannot use insulin effectively. As a result, blood glucose levels rise dangerously. Over time, high blood sugar damages nerves, kidneys, eyes, and the heart.
| 📊 Global Prevalence: 537 million adults affected worldwide (IDF, 2021) |
| 📊 Annual New Cases: Over 3 million new diagnoses per year in the US alone (CDC, 2023) |
| 📊 Insulin Dependence: Approximately 25–30% of type 2 diabetics eventually require insulin therapy |
| 📊 Adherence Problem: Up to 40% of patients stop daily insulin within 1 year due to injection burden |
| 📊 Global Impact: T2D accounts for over 90% of all diabetes cases worldwide |
Moreover, many patients delay starting insulin — a problem known as ‘psychological insulin resistance’ — simply because of the daily injection burden. Consequently, blood sugar stays uncontrolled for too long. Therefore, a once-weekly insulin option like Awiqli (insulin icodec-abae) could transform how patients manage their condition.
What Is Awiqli (Insulin Icodec)?
Awiqli (insulin icodec-abae) is a long-acting basal insulin analogue manufactured by Novo Nordisk. It is available as a 700 units/mL injectable solution, administered subcutaneously once weekly using the Awiqli FlexTouch prefilled pen. Furthermore, it is the first and only FDA-approved once-weekly basal insulin for adults with type 2 diabetes.
Additionally, Awiqli is now approved in the US, EU, Canada, Australia, Japan, and 13 further countries. It is not currently approved for type 1 diabetes. The FDA previously raised concerns about hypoglycaemia risk in type 1 patients.
How Does Awiqli (Insulin Icodec) Work?
Mechanism of Action of Insulin Icodec-Abae
Unlike daily basal insulins, insulin icodec-abae was specifically engineered for once-weekly dosing. Think of it as a ‘slow-release reservoir’ of insulin. After each weekly injection, icodec binds reversibly to albumin — a protein in the blood. This binding keeps it circulating in the bloodstream for up to seven days, releasing active insulin gradually and consistently.
| 🔬 HOW AWIQLI WORKS — Step by Step: |
| Step 1 → Injected subcutaneously once per week via FlexTouch pen |
| Step 2 → Insulin icodec binds reversibly to albumin in the bloodstream |
| Step 3 → Gradual, sustained release of active insulin over 7 days |
| Step 4 → Active insulin stimulates glucose uptake by cells and suppresses liver glucose production |
| Step 5 → Blood sugar levels remain stable throughout the entire week |
Therefore, Awiqli provides the same steady basal insulin coverage as daily insulins — but requires only one injection per week instead of seven. As a result, patients experience a dramatically reduced injection burden.
Key Benefits and Clinical Evidence
Why Doctors Prescribe Awiqli (Insulin Icodec)
The approval of Awiqli is based on the ONWARDS Phase 3a clinical programme — four large, randomised, active-controlled trials involving approximately 2,680 adults with uncontrolled type 2 diabetes. Each trial compared once-weekly icodec against a daily basal insulin.
Here are the key clinical benefits supported by trial data:
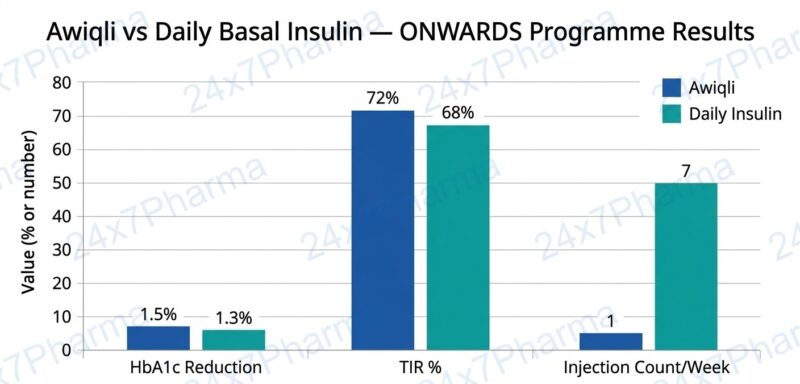
- ✅ Superior HbA1c Reduction: In ONWARDS 1, icodec reduced HbA1c from 8.50% to 6.93%, versus 7.12% with glargine U100 (difference: –0.19%; p<0.05) — published in The New England Journal of Medicine (NEJM, 2023)
- ✅ Significant Injection Reduction: Awiqli cuts weekly injections from 7 (daily glargine) to just 1 — a 86% reduction in injection events per year
- ✅ Consistent Efficacy Across Combinations: Proven effective alongside mealtime insulin, oral antidiabetics (e.g. metformin), and GLP-1 receptor agonists in four separate trials
- ✅ Improved Time in Range (TIR): In one trial, Awiqli achieved 62.34% TIR vs 59.93% with insulin degludec — the first FDA label to include TIR metrics
- ✅ Comparable Safety Profile: Overall adverse event rates were consistent with daily basal insulins across all four ONWARDS trials
Dosage and Administration of Awiqli
Always follow your endocrinologist’s or physician’s prescribed dosage. Do not self-adjust. The table below summarises standard administration guidance:
| Parameter | Details |
|---|---|
| Standard Dose | 700 units/mL — dose calculated per patient by physician |
| Frequency | Once weekly — same day each week |
| Administration | Subcutaneous injection via Awiqli FlexTouch pen |
| With or Without Food | May be administered with or without food |
| Missed Dose | If > 4 days remain before next dose, inject as soon as possible. If ≤ 4 days remain, skip and resume on next scheduled day |
| Dose Adjustment (Renal) | Use with caution in severe renal impairment — monitor glucose closely |
| Dose Adjustment (Hepatic) | Use with caution in hepatic impairment — increased hypoglycaemia risk |
| Storage | Before first use: refrigerate (2°C–8°C). After first use: store at room temperature below 30°C for up to 8 weeks |
| Approved Device | Awiqli FlexTouch prefilled pen — U-700 formulation |
⚠️ Important Note: When switching from daily basal insulin to Awiqli, closely monitor blood glucose during the first weeks. Your doctor will calculate the conversion dose based on your current regimen.
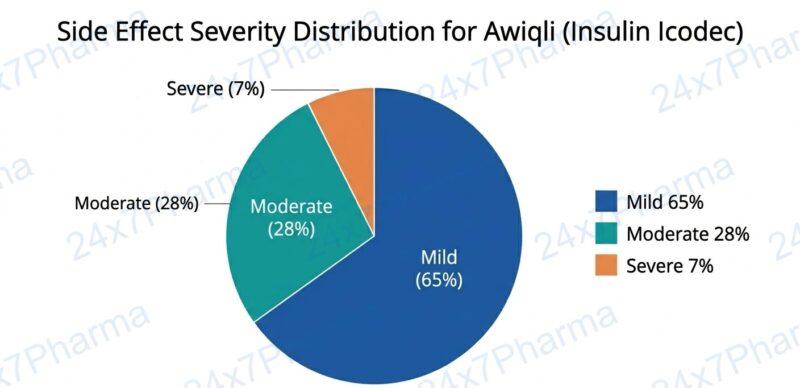
Side Effects of Awiqli (Insulin Icodec)
As with all insulin therapies, Awiqli carries the risk of certain side effects. However, the overall safety profile observed across the ONWARDS programme was consistent with the daily basal insulin class.
| ⚠️ Common Side Effects (>10% of patients) | 🚨 Serious Side Effects (seek immediate care) |
|---|---|
|
|

Who Should (and Should Not) Take Awiqli?
Awiqli is approved for adults with type 2 diabetes who require basal insulin therapy. In addition, it is suitable for patients already using daily basal insulin who want a more convenient once-weekly option.
Ideal Patient Profile
- Adults aged 18+ with type 2 diabetes (T2D) — not type 1
- Patients who struggle with daily injection adherence
- Patients using once-weekly GLP-1 medications (compatible with Awiqli)
- Patients on oral antidiabetics who need insulin escalation
Who Must NOT Take Awiqli
- Contraindicated: Patients with known hypersensitivity to insulin icodec-abae or any component of the formulation
- Not Approved For: Type 1 diabetes (T1D) — FDA specifically excluded this indication due to hypoglycaemia risk
- Use with Caution: Severe renal or hepatic impairment — increased risk of hypoglycaemia; frequent glucose monitoring required
- Pregnancy: Consult your specialist — adequate data not yet available for Awiqli during pregnancy or breastfeeding
Always consult your specialist or endocrinologist before starting Awiqli insulin icodec. Do not switch from your current insulin without medical supervision.
Patient Tips and Lifestyle Advice
- Pick one day, stick to it: Administer Awiqli on the same day every week. Consistency maintains stable blood glucose throughout the week.
- Monitor your blood sugar regularly: Especially in the first 4 weeks after starting. Keep a glucose diary to share with your doctor at each review.
- Eat a balanced diet: Insulin works best with consistent carbohydrate intake. Avoid sudden fasting or bingeing, which can trigger hypoglycaemia.
- Stay hydrated: Drink at least 6–8 glasses of water daily. Dehydration can affect insulin absorption and glucose control.
- Watch for hypo symptoms: Shakiness, sweating, dizziness, and confusion signal low blood sugar. Keep glucose tablets or juice on hand at all times.
- Avoid thiazolidinediones if possible: These oral diabetes drugs increase fluid retention and can worsen oedema when combined with insulin. Discuss with your doctor.
- Store the pen correctly: Before first use, refrigerate. After first use, store at room temperature below 30°C. Use within 8 weeks.
- Never mix Awiqli with other insulins: Do not dilute or mix Awiqli in the same syringe or pen with any other insulin preparation.
Frequently Asked Questions (FAQ)
Q: How long does Awiqli take to work for type 2 diabetes?
A: Awiqli begins working within hours of the first injection. However, stable blood glucose control typically develops over 2–4 weeks as the drug reaches a steady state. Your doctor will monitor your HbA1c at 3 months.
Q: Can I take Awiqli with food?
A: Yes. Unlike oral diabetes medicines that require specific food timing, Awiqli can be injected with or without food. Administer it subcutaneously on the same day each week, regardless of meals.
Q: What should I do if I miss a dose of Awiqli?
A: If more than 4 days remain before your next scheduled dose, inject as soon as you remember. If 4 days or fewer remain before your next dose, skip the missed dose and resume your normal weekly schedule. Never double dose.
Q: Are there alternatives to Awiqli for type 2 diabetes requiring insulin?
A: Yes. Daily basal insulins such as insulin glargine (Lantus) and insulin degludec (Tresiba) are established alternatives. Awiqli’s unique advantage is its once-weekly dosing. Consult your endocrinologist to determine which is right for you.
Conclusion
Awiqli (insulin icodec-abae) is a landmark advancement in diabetes care. Furthermore, it is the world’s first once-weekly basal insulin for type 2 diabetes — proven across four Phase 3 trials. In addition, it reduces the injection burden from seven daily shots to just one per week, improving patient adherence and quality of life. Therefore, for adults with type 2 diabetes who struggle with daily insulin, Awiqli offers a clinically proven, FDA-approved alternative.
⚠️ DISCLAIMER
The information in this blog is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting, stopping, or changing any medication.