Every year, approximately 1.3 million people worldwide acquire HIV — a virus that still has no cure (UNAIDS, 2023). However, a remarkable new tool is changing the prevention landscape. Lenacapavir, a first-in-class HIV capsid inhibitor, now offers twice-yearly injection-based pre-exposure prophylaxis (PrEP). Clinical trials show near-complete protection in high-risk populations — making this one of the most significant breakthroughs in HIV prevention history.
📊 Key Global HIV Statistics (UNAIDS 2023 / WHO 2024)
Daily oral PrEP adherence rates fall below 50% in many real-world settings — driving the urgent need for long-acting alternatives.
What Is HIV and Why Does Prevention Matter?
HIV (Human Immunodeficiency Virus) attacks the immune system — specifically CD4 T-cells, which are essential for fighting infections. Without treatment, HIV can progress to AIDS (Acquired Immunodeficiency Syndrome), leaving people vulnerable to life-threatening opportunistic infections. HIV disproportionately affects communities with limited healthcare access, making prevention an urgent global priority.
Modern PrEP medicines — including lenacapavir — provide powerful tools to stop transmission before it occurs. While standard daily oral PrEP (tenofovir-based) is effective when taken consistently, adherence remains a significant barrier. Lenacapavir, administered just twice a year, directly addresses this critical unmet need.
💊 Browse 24x7Pharma’s full range of HIV prevention and treatment medicines at our HIV Infection category page →
What Is Lenacapavir? A New Class of HIV Medicine
Lenacapavir (brand name Sunlenca) is the world’s first HIV-1 capsid inhibitor approved for clinical use. Developed by Gilead Sciences, it works differently from all previous antiretroviral drug classes.
In 2022, the FDA and EMA approved lenacapavir for heavily treatment-experienced people with multidrug-resistant HIV. Then in 2024, breakthrough clinical trial results demonstrated its exceptional efficacy as twice-yearly PrEP.
Lenacapavir is notable for its extremely long half-life — enabling the every-six-month dosing schedule. As the first of a new drug class, it also retains activity against strains resistant to all other drug classes.
How Does Lenacapavir Work?
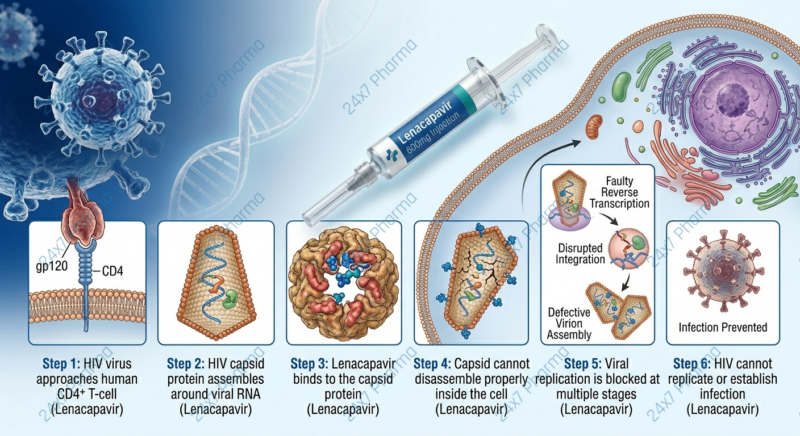
Mechanism of Action — Capsid Inhibitor
The Capsid
The HIV capsid is a molecular “armoured vehicle” that carries the virus’s genetic blueprint into human cells.
Lenacapavir Locks It
Lenacapavir binds to the capsid protein and blocks multiple stages of HIV replication simultaneously.
Multi-Stage Protection
Unlike single-stage inhibitors, lenacapavir is effective even against HIV strains resistant to older drug classes.
Its unique mechanism also enables the ultra-long half-life that supports twice-yearly dosing — something no previous HIV medicine achieved.
Clinical Trial Results
Key Benefits & Clinical Evidence
PURPOSE 1 — Sub-Saharan Africa (Young Women)
100% Efficacy
Zero HIV infections in the lenacapavir group versus a 2.0% annual incidence in the background population. (NEJM, 2024)
PURPOSE 2 — Global Diverse Populations
96% Reduction in HIV Incidence
Compared to daily oral tenofovir alafenamide/emtricitabine (TAF/FTC) PrEP. Effective in cisgender women, MSM, transgender women, and people who inject drugs.
2 injections/year
vs 365 daily pills
Broad eligibility
diverse populations
Favourable safety
no major systemic concerns
Lenacapavir Dosage & Administration
The dosing schedule for lenacapavir as PrEP is straightforward. However, injections must be administered by a trained healthcare professional — not self-administered at home.
⚠️ Important: Always follow your doctor’s or pharmacist’s prescribed schedule. Do not attempt to self-adjust the injection timing without medical guidance.
Lenacapavir Side Effects — What to Expect
Like all medicines, lenacapavir may cause side effects. However, the overall safety profile observed in clinical trials was favourable and most people tolerate it well.
⚡ Common Side Effects
- 💉 Injection site reactions — nodules, pain, induration (mild to moderate)
- 🟣 Injection site bruising — temporary, resolves on its own
- 🤢 Nausea — mild, generally transient
- 🤕 Headache — reported in a minority of participants
🚨 Seek Immediate Medical Attention
- 😮 Allergic reaction — rash, facial/throat swelling, difficulty breathing
- 🔴 Severe injection site complications — significant swelling, warmth, redness suggesting infection
- 🟡 Liver symptoms — yellowing of skin or eyes, dark urine, severe abdominal pain
Who Should — and Should Not — Consider Lenacapavir PrEP?
✅ Ideal Candidates
- HIV-negative adults at substantial risk of HIV acquisition
- Individuals who struggle with daily oral PrEP adherence
- People in high-prevalence regions where 6-month clinic visits are feasible
- Those who have tried oral PrEP and experienced adherence challenges
🚫 Not Suitable For
- People already living with HIV (without specialist assessment)
- Known hypersensitivity to lenacapavir or any component
- Pregnancy or breastfeeding — consult a specialist (emerging safety data)
- Self-administration at home — must be given by a healthcare professional
Patient Tips & Lifestyle Advice
Keep Every Appointment
Set calendar reminders 6 months in advance. Missing an injection significantly reduces protection.
Regular HIV Testing
Get tested for HIV before each injection. If infection occurs, lenacapavir must not be used as sole treatment without specialist guidance.
STI Screening
PrEP reduces HIV risk but does not protect against other sexually transmitted infections. Regular STI testing is important.
Drug Interactions
Inform your doctor of all medicines, supplements, and herbal products you take, as interactions are possible.
Report Reactions Promptly
Mild nodules at the injection site are expected. Report severe pain, warmth, or spreading redness to your clinic immediately.
General Health
Maintain a balanced diet and stay well hydrated. A healthy immune system supports overall wellbeing during PrEP.
Frequently Asked Questions
Conclusion
A New Era of HIV Prevention Has Arrived
Lenacapavir represents a genuine paradigm shift. As a first-in-class capsid inhibitor delivered just twice a year, it removes the daily pill burden that limits oral PrEP for millions. With PURPOSE 1 showing 100% efficacy and PURPOSE 2 showing a 96% reduction in HIV incidence, public health experts are optimistic that lenacapavir could dramatically reduce global HIV transmission if access is scaled appropriately.
Disclaimer: The information in this blog is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting, stopping, or changing any medication. 24x7Pharma does not diagnose, prescribe, or replace professional medical guidance.
© 2026 24x7Pharma. International Online Pharmacy. Educational purposes only. Not medical advice.
support@24x7pharma.com | www.24x7pharma.com