Lifyorli (Relacorilant): The World’s First Cortisol-Blocking Cancer Drug That Makes Chemotherapy Work Better in Ovarian Cancer
Introduction
Ovarian cancer is one of the most challenging gynaecological malignancies worldwide, with the International Agency for Research on Cancer (IARC) reporting over 324,000 new cases and more than 206,000 deaths annually. For women whose disease has stopped responding to platinum-based chemotherapy — a condition known as platinum-resistant ovarian cancer — treatment options have historically been limited and outcomes poor.
In this guide, we explore how Lifyorli (relacorilant) — a groundbreaking glucocorticoid receptor (GR) antagonist and the world’s first cortisol-blocking agent approved specifically to enhance cancer chemotherapy — works, its clinical benefits, correct dosage, and side effects.
What Is Platinum-Resistant Ovarian Cancer?
Ovarian cancer arises from the cells lining the ovaries or fallopian tubes and is frequently diagnosed at an advanced stage when it has already spread. The standard first-line treatment is platinum-based chemotherapy (carboplatin or cisplatin), which is effective for most women initially. However, for a significant proportion, the cancer eventually stops responding — or never responds — to platinum drugs. This is defined as platinum-resistant ovarian cancer (PROC), a condition carrying a particularly poor prognosis.
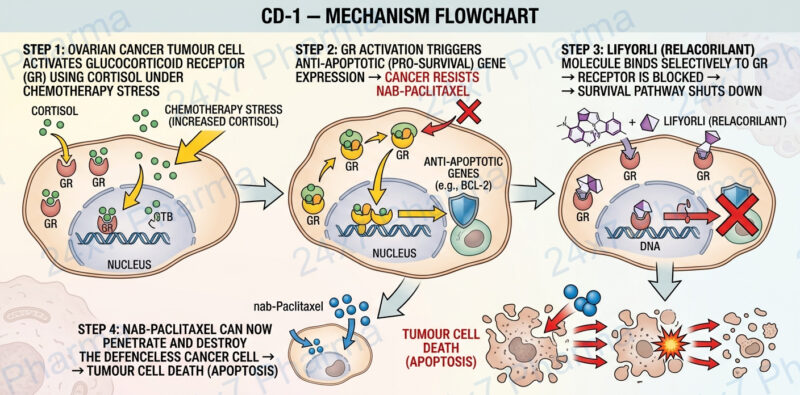
Emerging research has revealed a critical biological driver behind this resistance: elevated cortisol activity. Tumour cells activate glucocorticoid receptors in response to stress hormones, using this pathway as a shield against chemotherapy. Standard treatments often fail to address this resistance mechanism, highlighting the urgent need for targeted platinum-resistant ovarian cancer treatment options.
| 📊 STATISTICS CALLOUT BOX — Ovarian Cancer at a Glance |
|---|
| 📊 Global Prevalence: Approx. 1.3 million women living with ovarian cancer worldwide (IARC, 2023) |
| 📊 Annual New Cases: Over 324,000 new diagnoses globally per year (IARC, 2022) |
| 📊 5-Year Survival Rate: ~49% overall; drops to ~30% for advanced-stage disease (SEER, NCI) |
| 📊 Platinum Resistance Rate: ~25% of patients are primary platinum-resistant; recurrence brings resistance in up to 70% (ESMO Guidelines) |
| 📊 Countries Most Affected: USA, UK, China, India, Germany, Australia (highest absolute case volumes globally) |
The arrival of Lifyorli as a cortisol-blocking partner to chemotherapy represents the first therapy to directly target this resistance pathway, offering new hope for patients who have exhausted platinum options.
What Is Lifyorli (Relacorilant)?
Lifyorli is the brand name for relacorilant, a first-in-class selective glucocorticoid receptor (GR) antagonist developed by Corcept Therapeutics. It is the first drug of its kind approved by the U.S. Food and Drug Administration (FDA) specifically for use in combination with chemotherapy for platinum-resistant ovarian cancer. The FDA granted approval in early 2025, marking a historic milestone in oncology.
Unlike traditional chemotherapy drugs that directly target tumour DNA, Lifyorli works at the hormonal level — blocking the cortisol receptor that cancer cells weaponise to escape destruction. It is available as an oral tablet, making it a convenient addition to an existing chemotherapy regimen.
How Does Lifyorli Work?
Mechanism of Action of Relacorilant
To understand Lifyorli, think of glucocorticoid receptors as a secret backdoor that cancer cells use to protect themselves. Under stress — including the stress of chemotherapy — the body releases cortisol. Ovarian cancer cells use cortisol to activate anti-survival signals that make them harder to kill. Lifyorli acts as a precision lock on that backdoor: it binds tightly and selectively to the glucocorticoid receptor (GR), preventing cortisol from activating cancer’s protective programmes.
When Lifyorli blocks GR, cancer cells lose their cortisol-driven survival advantage. This makes them significantly more vulnerable to nab-paclitaxel, the chemotherapy partner drug, which can then destroy tumour cells far more effectively.
Key Benefits & Clinical Evidence of Lifyorli
Why Doctors Prescribe Lifyorli
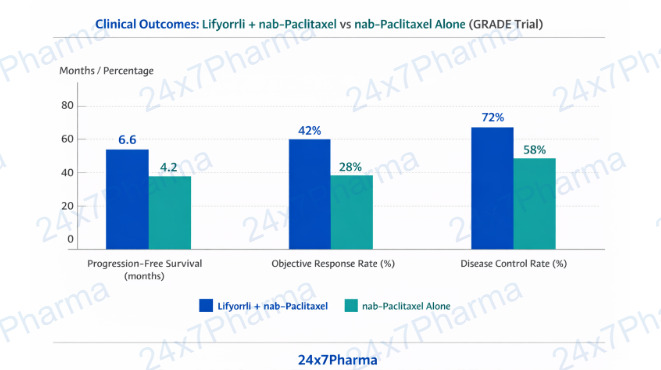
The clinical case for Lifyorli rests on the landmark GRADE clinical trial, a Phase 3 randomised, double-blind study evaluating relacorilant + nab-paclitaxel vs nab-paclitaxel alone in platinum-resistant ovarian cancer. The headline result: progression-free survival (PFS) was significantly improved in the combination arm, with a hazard ratio demonstrating a meaningful reduction in the risk of disease progression or death.
- First-in-class mechanism: The only FDA-approved glucocorticoid receptor antagonist for cancer — a truly novel approach.
- Improved progression-free survival: GRADE trial demonstrated statistically significant PFS benefit vs chemotherapy alone (HR ~0.66, per published data).
- Enhanced objective response rate (ORR): Higher tumour response rates observed in patients receiving the combination regimen.
- Oral, convenient dosing: Administered as a once-daily tablet — integrates seamlessly into existing chemotherapy cycles.
- Targets the resistance mechanism directly: Addresses the cortisol-driven resistance biology rather than simply adding more cytotoxic therapy.
For patients exploring platinum-resistant ovarian cancer treatment options, Lifyorli represents the most scientifically advanced option currently available.
Lifyorli Dosage & Administration
| Parameter | Details |
|---|---|
| Standard Dose | Relacorilant 100 mg orally once daily |
| Frequency | Once daily (continuous dosing) |
| Duration | Throughout nab-paclitaxel chemotherapy cycles |
| With or Without Food | Can be taken with or without food; consistent timing recommended |
| Missed Dose Instructions | Take as soon as remembered on the same day. Do not double dose the next day. |
| Dose Adjustment (Renal) | No significant adjustment required for mild-to-moderate renal impairment; consult physician for severe cases |
| Dose Adjustment (Hepatic) | Use with caution in hepatic impairment; avoid in severe hepatic impairment |
| Storage Conditions | Store at room temperature (20–25°C), away from moisture and direct light |
| ⚠️ Important: Always follow your oncologist’s prescribed dosage schedule. Do not self-adjust the dose of Lifyorli or nab-paclitaxel without medical guidance. |
|---|
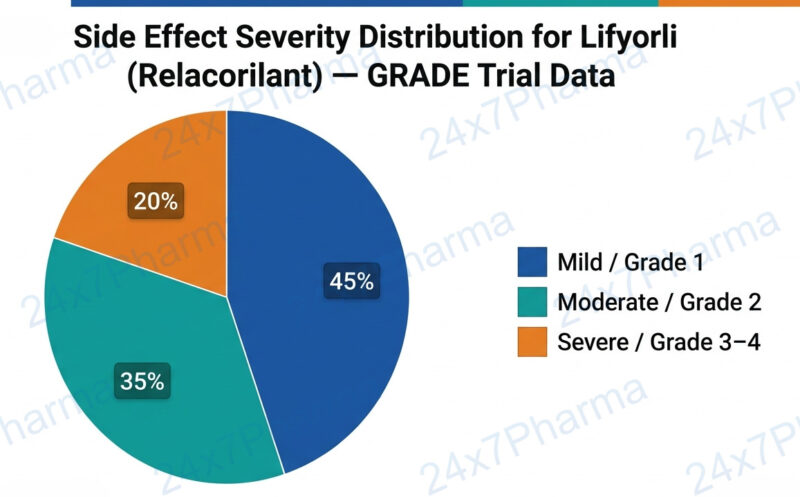
Side Effects of Lifyorli (Relacorilant)
| ⚠️ Common Side Effects (>10% patients) | 🚨 Serious Side Effects (seek immediate care) |
|---|---|
| • Fatigue and weakness
• Nausea and vomiting • Peripheral neuropathy (numbness/tingling) • Alopecia (hair loss) • Decreased appetite |
• Severe adrenal insufficiency
• Significant QT interval prolongation • Severe hypersensitivity / allergic reaction |
Who Should (and Should Not) Take Lifyorli?
Lifyorli is indicated for adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer who have received one to three prior lines of therapy and are being treated with nab-paclitaxel. Biomarker testing is not required, but patients should have adequate organ function.
Who must NOT take Lifyorli: Patients with known hypersensitivity to relacorilant or any component of the formulation should not take this medicine. Concurrent use of strong CYP3A inhibitors or inducers requires careful physician assessment. Lifyorli is contraindicated in pregnancy and must not be used by women who are breastfeeding. Patients with severe hepatic impairment (Child-Pugh C) should avoid use.
Always consult your specialist or oncologist before starting Lifyorli to confirm it is appropriate for your specific disease profile and treatment history.
Patient Tips & Lifestyle Advice for Lifyorli Users
- Stay consistently hydrated: Drink at least 8 glasses of water daily. Good hydration helps reduce nausea and fatigue and supports kidney function during chemotherapy.
- Eat small, frequent meals: Nausea is common with nab-paclitaxel combinations. Light, easily digestible foods taken at regular intervals can help maintain appetite and energy.
- Monitor for neuropathy: Report any new tingling, numbness, or weakness in hands or feet to your oncologist promptly, as peripheral neuropathy may require dose adjustment.
- Missed dose: If you miss a dose of Lifyorli, take it as soon as you remember on the same day. Do not take a double dose the following day to compensate.
- Drug interactions to avoid: Avoid grapefruit and grapefruit juice, strong CYP3A4 inhibitors (e.g., ketoconazole, clarithromycin), and strong CYP3A4 inducers (e.g., rifampicin, St. John’s Wort). Always inform your doctor of all medications, including supplements.
- When to call your doctor immediately: Seek urgent care if you experience severe fatigue, signs of infection (fever >38°C), unusual bleeding, chest pain, shortness of breath, or signs of adrenal crisis (extreme dizziness, weakness, low blood pressure).
- Adherence is critical: Take Lifyorli at the same time each day to maintain stable blood levels. Use a pill organiser or phone reminder to stay on schedule.
- Use effective contraception: Women of childbearing potential must use highly effective contraception during treatment and for a period after completing therapy, as Lifyorli can cause foetal harm.
Frequently Asked Questions (FAQ)
Q1: How long does Lifyorli take to work for platinum-resistant ovarian cancer?
Lifyorli works alongside nab-paclitaxel chemotherapy from the first cycle. Clinical response (tumour shrinkage) is typically assessed after 2–3 cycles (6–9 weeks) via imaging. Your oncologist will monitor your response through regular CT scans and tumour markers such as CA-125.
Q2: Can Lifyorli be taken with food?
Yes — Lifyorli can be taken with or without food. However, consistency is important: choose a time of day and stick with it. Avoid taking it with grapefruit juice or foods high in grapefruit, as these can interfere with how the drug is metabolised.
Q3: What should I do if I miss a dose of Lifyorli?
If you miss a dose, take it as soon as you remember — but only on the same day. If you don’t remember until the following day, skip the missed dose entirely and resume your normal schedule. Never take two doses in one day.
Q4: Are there any alternatives to Lifyorli for platinum-resistant ovarian cancer?
Prior to Lifyorli, options for platinum-resistant ovarian cancer included single-agent chemotherapy agents (gemcitabine, topotecan, liposomal doxorubicin), bevacizumab combinations, and PARP inhibitors (for BRCA-mutated tumours). Lifyorli is currently the only FDA-approved GR antagonist combination for this indication, representing a uniquely differentiated mechanism compared to all alternatives.
Conclusion
Lifyorli (relacorilant) represents one of the most meaningful advances in ovarian cancer treatment in recent years — the first drug to block the cortisol pathway that cancer cells use to resist chemotherapy, and the first of its class approved by the FDA. For women battling platinum-resistant ovarian cancer, it offers a scientifically grounded, clinically proven combination option that meaningfully extends progression-free survival.
Whether you are a patient, caregiver, or healthcare provider seeking more information, Lifyorli stands as a proven, trusted advance in ovarian cancer treatment options — backed by Phase 3 trial evidence and FDA approval.
Disclaimer
| The information in this article is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting, stopping, or changing any medication. |
© 2026 24x7Pharma. International Online Pharmacy. Educational purposes only. Not medical advice.
support@24x7pharma.com | www.24x7pharma.com